First Total Synthesis of Glaucocalyxin A (Y. Jia, 2020)
Using a radical cyclization strategy at the very beginning of their synthesis, the Yanxing Jia (贾彦兴) research group from Peking University could successfully report the first total synthesis of (−)-glaucocalyxin A (1). The synthesis of the highly oxidated diterpenoid was reported in the journal Angewandte Chemie. From a retrosynthetic point of view (see Figure 1) the natural product should be available from 2 by late-stage functionalization. The precursor itself should be synthesized using Diels Alder cyclization from polyene 3. This substrate is available from 4 by aldol addition. As mentioned before, a radical and oxidative cyclization should set the bicyclo[3.2.1]octane system at the very beginning starting from 5.
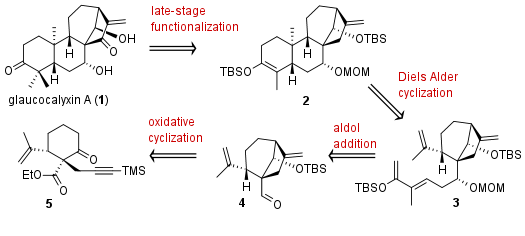 Figure 1: Retrosynthetic analysis for the total synthesis of glaucocalyxin A (1).
Figure 1: Retrosynthetic analysis for the total synthesis of glaucocalyxin A (1).
Radical Cyclization for the Construction of the bicyclooctane System
Starting with unsaturated ketone 6 (see Scheme 1) enantioselective Michael addition with a chiral copper ligand followed by trapping the enolate with NCCO2Et yielded in 7. Note, that only in this very first step a chiral catalyst was used. All further chiral centers were established by substrate control. The Jia Group then treated dicarbonyl 7 with base and α alkylation was followed by acidic TMS deprotection to get cyclization precursor 5 in over 50% overall yield. The following cyclization was optimized using a model substrate (not shown) to give the desired bicyclo-[3.2.1]-octance system in 53% yield. TMS deprotection then liberated the terminal alkene 8. Using common methods, the ketone was reduced and TBS protected. Then DIBAlH reduction and DMP oxidation gave aldehyde 4 in 72% yield over 4 steps. The author described that it was not possible to set the desired stereocenter during the ketone reduction, which therefore has to been set in a later sequence. Using a vinylogous aldol addition finally liberated unsaturated ester 9 in excellent yield but moderate diastereoselectivity.
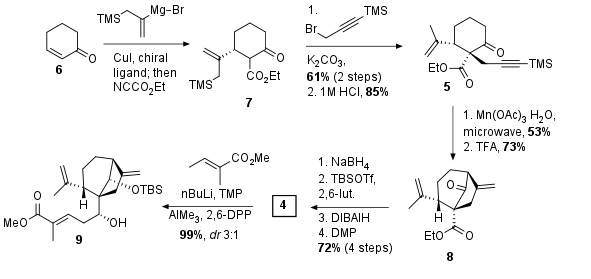 Scheme 1: Radical cyclization and further transformation led to fragment 9.
Scheme 1: Radical cyclization and further transformation led to fragment 9.
Completion of the Total Synthesis
With fragment 9 in hand, the Jia group first protected the alcohol with MOM and then used a two-step procedure to transform the ester into the methyl ketone using the Weinreb amide 10. Then the formation of the silyl enol ether was required to initiate the following Diels Alder cyclization. The cyclization under thermal conditions delivered smoothly 2 as a single isomer in excellent yield. The silyl enol ether was carefully cleaved and in the presence of methyl iodide under strongly anhydrous conditions the second geminal methyl group could be installed. The following ketone reduction and acetate protection then yielded in 11. Now a three-step protocol was necessary for epimerization of the aforementioned undesired configuration. First deprotection and oxidation with DMP gave the ketone in two steps. Then Birch reduction finally led to the desired configuration together with acetate cleavage to 12. To complete the total synthesis, allylic oxidation was followed by DMP oxidation and MOM deprotection to gave the desired glaucocalyxin in three steps with 33% yield.
 Scheme 2: Completion of the total synthesis.
Scheme 2: Completion of the total synthesis.
With this approach, the Jia group could successfully synthesize more than 100 mg of the diterpenoid. Noteworthy, they could also provide a more than complete set of analytical data which includes mass spectra, optical rotation, IR spectra, melting point analysis, and the comparison of NMR signals of the synthetic material with the natural product in different deuterated solvents. This should be a friendly reminder for all synthetic chemists in the field that it is actually possible to obtain detailed analytical data even after a 24 step synthesis.
Published in J. Guo, B. Li, W. Ma, M. Pitchakuntla, Y. Jia Angewandte Chemie Int. Ed. 2020, 59, 15195-15198. doi: 10.1002/anie.202005932

No comments to display
No comments to display